Is there such a thing as a molecular organic alloy?
Abstract
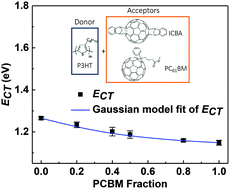
In inorganic materials, an alloy is a mixture of two or more substances that generally exhibits electronic and/or physical properties that differ from those of its constituents. In organic systems, the formation of a “molecular alloy” comprising mixtures of molecular organic materials has also been proposed. We test the validity of this concept via the study of the optoelectronic properties of a ternary system that has previously been identified to form a molecular acceptor alloy, namely a blend of a poly(3-hexylthiophene) (P3HT) donor, with two acceptors indene-C60 bisadduct (ICBA) and phenyl-C61-butyric acid methyl ester (PC61BM) [R. A. Street, et al., J. Am. Chem. Soc., 2013, 135, 986–989]. Using photoelectron spectroscopy, we find that the ICBA:PC61BM blend shows the same highest occupied molecular orbital and exciton energies as that of ICBA, indicating the absence of a new exciton state in the blend. Furthermore, charge transfer state spectra of ternary blends are found to comprise a simple linear superposition of the corresponding binaries. From these results, no evidence of new, emergent electronic states is found to support the existence of a molecular alloy in this system. To our knowledge there is as yet no clear evidence of the existence of an alloy in any organic semiconductor system. We discuss the criteria that should be met by a molecular organic alloy and procedures needed for their unambiguous identification.


 Please wait while we load your content...
Please wait while we load your content...