Nanothin film conductivity measurements reveal interfacial influence on ion transport in polymer electrolytes†
Abstract
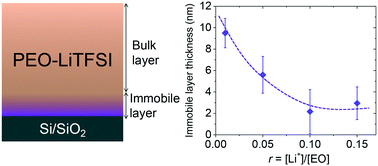
The interfacial region where ion-transporting polymer chains are anchored to a hard, insulating phase is a major factor dictating the limits of ion-conduction in nanostructure-forming electrolytes. In this work, we investigate the effect of an end-grafted poly(ethylene oxide) (20 kg mol−1) surface on the ionic conductivity σ of PEO and lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) salt mixtures. Specifically, we characterize nanothin films in the range of ca. 10 to 250 nm, which amplify the contributions from the polymer/substrate interface that dictate any deviations from expected bulk conductivity σbulk values. Conductivity measurements reveal a monotonic decrease in σ upon decreasing film thickness at all values of r (r = molar ratio of Li+ to EO units). The reduction from bulk-like σ occurs for film thicknesses approximately 100 nm and below for all values of r. This trend in conductivity arises from the presence of the underlying grafted-PEO layer. Through a thickness dependence normalized conductivity study, we observe nanoscale constraints leading to deviation from intrinsic conductivity of bulk PEO–LiTFSI electrolytes. These nanoscale constraints correspond to an immobile interfacial zone whose thickness hint ranges from 9.5 ± 1.4 nm at r = 0.01 to 2.9 ± 1.5 nm at r = 0.15 in our nanothin films that impedes ion transport. Overall, we have presented a robust platform that facilitates probing the role of polymer-grafted surfaces on the σ of polymer electrolytes.
- This article is part of the themed collection: Charge Transporting Nanostructured Polymers for Electrochemical Systems


 Please wait while we load your content...
Please wait while we load your content...