Macrocyclic host appended carbon nanotubes for selective adsorption of metal ions: combined experimental, DFT and MD studies†
Abstract
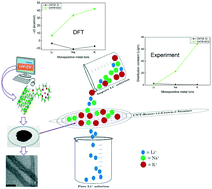
Crown ethers are very useful for metal ion recognition due to their nanocavity based specific ion selectivity, which on functionalization with carbon nanotubes (CNTs) can be employed as specific metal ion filters by exploiting their different interactions with metal ions. The present study focuses on designing new functionalized nanomaterials using density functional theory and molecular dynamics simulations for understanding their interactions with metal ions of interest followed by synthesis and adsorption experiments. From the analysis of computational results, it is seen that the K+ ion binds strongly to the benzo-15-crown-5 (B15C5) ether and B15C5 appended CNT (CNT-B15C5) followed by Na+ and Li+. Both theoretical and experimental results showed that the adsorption capacity of CNT-B15C5 is much higher than that of free CNTs reflecting the cooperative effect of the π-electron of the CNT along with the dipole of “O” donors of the crown ether. The adsorption ability of the CNT has been enhanced remarkably by more than 7500% for K+ after functionalization with B15C5. The experimental result reveals zero Li+ ion adsorption by CNT-B15C5. Further, MD simulation also confirmed the selective inclusion of the K+ ion within the cavity of the crown ether in the presence of Li+ and Na+ ions. We anticipate that the functionalized CNT-B15C5 can be used for preparing high purity lithium compounds by selectively filtering K+ and Na+ ions and also for other chemically and biologically important ion filter/channels.


 Please wait while we load your content...
Please wait while we load your content...