Semi-quantitative design of black phosphorous field-effect transistor sensors for heavy metal ion detection in aqueous media†
Abstract
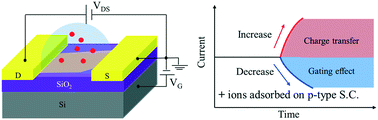
Two-dimensional (2D) crystalline nanomaterial based field-effect transistor (FET) water sensors are attracting increased attention due to their low cost, portability, rapid response, and high sensitivity to aqueous contaminants. However, a generic model to aid in sensor design by describing direct interactions between metal ions and 2D nanomaterials is lacking. Here, we report a broadly applicable statistical thermodynamics model that describes the behavior of FET sensors (e.g., lower detection limit) by relying only on the ion concentration and intrinsic properties of the sensor material such as band gap and carrier effective mass. Two regimes of the sensing mechanism (charge transfer vs. electrostatic gating) were predicted, depending on the relative size of the Debye screening length in the sensor material and the distance between adsorbed ions. At a lower ion adsorption density, the charge transfer effect is dominant, while the evolution from charge transfer to electrostatic gating effect occurs at a higher adsorption density as the distance between adsorbed ions approaches the Debye length. Owing to its tunable band gap, black phosphorus (BP) nanosheet FET sensors were selected to semi-quantitatively validate the model including the predicted evolution between the two sensing regimes. Among Na+, Mg2+, Zn2+, Cd2+, Pb2+, and Hg2+ ions, BP nanosheet FET sensors were more responsive to Hg2+ ions for probe-free detection. The theoretical lower detection limit of Hg2+ ions can reach 0.1 nM (0.1 fM) in tap (deionized) water.
- This article is part of the themed collection: 2D materials: molecular design and engineering perspectives


 Please wait while we load your content...
Please wait while we load your content...