Aminopyrimidine–galactose hybrids are highly selective galectin-3 inhibitors†
Abstract
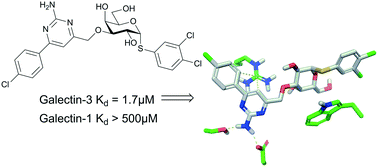
Galectins are a family of carbohydrate recognition proteins involved in, among other things, modulating cell signalling and cell–environment interactions, giving them roles in several pathologies like cancer and idiopathic lung fibrosis. Hence, developing new galectin inhibitors with high affinity and high selectivity is important to be able to target such diseases. Most existing galectin inhibitors have a disaccharide scaffold, but there has been success as of late in developing monogalactoside inhibitors such as α-arylthioglycosides. Here, we report aminopyrimidine-derivatised galactosides as good galectin-3 inhibitors with affinities down to 1.7 μM and a more than 300-fold selectivity over galectin-1. Mutant studies replacing Arg144 in galectin-3 with lysine and serine support the hypothesis that the binding of the derivatives involves interactions with Arg144. Molecular dynamics simulations converged to stable poses of the inhibitor aminopyrimidine moiety with polar interactions with Asp148 and Ser237, while the aryl-aminopyrimidine ring stacked onto the side chain of Arg144. Hence, combining an aminopyrimidine motif with a phenyl α-thiogalactoside motif offers an attractive route towards highly selective galectin-3 inhibitors.


 Please wait while we load your content...
Please wait while we load your content...