Novel carbohydrate modified berberine derivatives: synthesis and in vitro anti-diabetic investigation†
Abstract
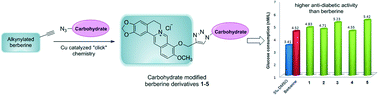
Berberine is a bioactive alkaloid used in Chinese medicine and has numerous positive effects on biological systems. This paper describes the facile and highly efficient synthesis of some carbohydrate modified berberine derivatives, and conjugation of the carbohydrate moiety with berberine was finished by “click” chemistry. The cytotoxicity and anti-diabetic measurements of all berberine derivatives were accomplished on HepG2 cell lines, and the results indicated that most of the derivatives exhibit higher anti-diabetic activity than berberine. The mannose modified berberine derivative has significantly lower cytotoxicity than berberine, and the induced IC50 value of this derivative is nearly 1.5 times that of berberine. Furthermore, this mannose modified berberine derivative exhibits high anti-diabetic activity at both high and low drug concentrations, thereby indicating its potential application for the development of novel anti-diabetic drugs.


 Please wait while we load your content...
Please wait while we load your content...