Single cell arrays of hematological cancer cells for assessment of lymphocyte cytotoxicity dynamics, serial killing, and extracellular molecules†
Abstract
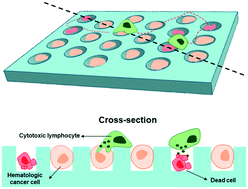
Cytotoxicity exerted by cytotoxic lymphocytes against cancer cells is an essential cellular function for successful cancer immunotherapy. Standard cytotoxicity assays mostly provide population level information, whereas live cell imaging-based cytotoxicity assays can assess single cell level heterogeneity. However, long term tracking of individual cytotoxic lymphocyte–hematological cancer cell interactions is technically challenging because both cells can float around and form multi-cellular aggregates. To overcome this limitation, single hematological cancer cell arrays with immobilized hematological cancer cells are fabricated using microwell arrays. Using this new platform, single cell level natural killer (NK) cell cytotoxicity against leukemic cells is quantitatively assessed. Depending on microwell surface adhesiveness and inter-microwell distances, distinct modes of NK–leukemic cell interactions that result in different NK cell cytotoxicity are observed. For microwell arrays coated with bovine serum albumin, which prevents cell adhesion, NK cells stably contacted cancer cells with rounded morphologies, whereas for microwell arrays coated with fibronectin (FN), which triggers integrin signals, NK cells contacting cancer cells exhibited dynamic behaviors with elongated morphologies and constantly explored extracellular spaces by membrane extension. In addition, FN on extracellular spaces facilitate NK cell detachment from leukemic cells after killing by providing anchorage for force transmission, and promote cytotoxicity and serial killing. Single hematologic cell arrays are not only an efficient method for lymphocyte cytotoxicity analysis but also a useful tool to study the role of signaling molecules in extracellular spaces on lymphocyte cytotoxicity.
- This article is part of the themed collection: Immunotherapy


 Please wait while we load your content...
Please wait while we load your content...