Determination of Mg isotope ratios without column chromatography for carbonates using sulphuric acid and MC-ICP-MS†
Abstract
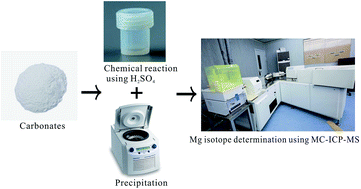
This study presents a practical method for high-precision Mg isotope determination without column chromatography for carbonate samples using sulphuric acid and MC-ICP-MS. The protocol uses the chemical reaction between carbonates and sulphuric acid to generate a CaSO4 precipitate and MgSO4 supernatant, which are used to determine Mg isotopic ratios. The matrix effects and the sulphuric acid conditions were rigorously examined using a Nu Plasma II in wet plasma mode. The precision and accuracy of this method were tested by measurements of the carbonite standard material JDo-1 over four months. The δ26Mg of JDo-1 is −2.32 ± 0.11‰ (2SD, n = 34) relative to DSM3, well consistent with the recommended value (−2.35 ± 0.15‰). The robustness of this method was further assessed by replicated analyses of ten Chinese carbonate reference materials that covered a large range of MgO contents from 5.98 wt% to 20.91 wt%.


 Please wait while we load your content...
Please wait while we load your content...