Calcium isotope determination in urine samples via the monitoring of 44CaF and 40CaF molecules by high-resolution continuum source graphite furnace molecular absorption spectrometry
Abstract
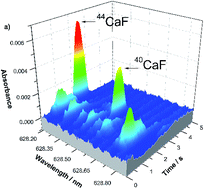
This study investigates the possibility of obtaining Ca isotopic information by monitoring the absorption spectrum of a diatomic molecule generated in a graphite furnace using a high-resolution continuum source spectrometer (HR CS GFMAS). The method proposed enables calcium isotopic analysis using CaF as the target species. The difference in wavelength between 40CaF (maximum at 628.534 nm) and 44CaF (maximum at 628.191 nm) absorption signals was found to be 337.8 pm, evidencing that both species can be determined simultaneously. The method developed relies on the determination of 40Ca and 44Ca concentrations based on calibration functions specific to each isotope and makes use of 12 μg of F as a molecule forming element without further chemical modifiers. The sensitivity of the method depends on the pH. It increases at pH values close to 2, and decreases until reaching a plateau at pH 6. The characteristic mass found for 40CaF was 0.11 μg for solutions with pH 2 and 0.18 μg for alkaline solutions. The respective limits of detection were 1 and 2 mg L−1. The method was applied to the analysis of three urine samples, spiked with different 40Ca and 44Ca amounts. Due to chlorine interference, before the analysis of urine, calcium must be separated from the samples by precipitation with ammonium oxalate. Recovery tests provided results between 95 and 109%, highlighting the potential of the method for 40Ca and 44Ca determination in tracer experiments.


 Please wait while we load your content...
Please wait while we load your content...