Time-resolved imaging of atoms and molecules in laser-produced uranium plasmas
Abstract
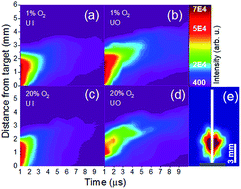
Gas-phase oxidation of uranium (U), plume chemistry, and the corresponding impact on optical emission features of the U plasma are investigated. Plasmas were produced via nanosecond laser ablation of a natural U target in a chamber where U oxidation was controlled by varying the oxygen partial pressure in an argon cover gas. Monochromatic imaging of U atoms and monoxide (UO) molecules was performed using narrow bandpass optical filters. Results reveal the spatio-temporal evolution of atomic and molecular species in the plasma. U oxides are found to be formed further from the target (in comparison to U atoms), where lower temperatures favor molecular recombination. Segregation between the distribution of U atoms and UO species is observed at later times of plasma evolution, and is more apparent at lower oxygen partial pressures. At higher oxygen partial pressures, significant variation in plume morphology is noticed for UO species, which can be attributed to the higher oxide (UxOy) formation further from the target. The monochromatic images of U atoms and UO molecules and corresponding spectral features at various oxygen partial pressures presented here provide unique insight into gas-phase, high-temperature U oxidation and chemistry, with implications for a wide range of nuclear applications, from stand-off detection of radioisotopes to forensics and safeguards.


 Please wait while we load your content...
Please wait while we load your content...