Thermally regulated molybdate-based ionic liquids toward molecular oxygen activation for one-pot oxidative cascade catalysis†
Abstract
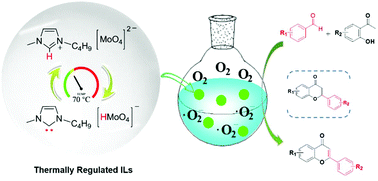
One-pot oxidative cascade catalysis plays a central role in the synthesis of key pharmaceutical and industrial molecules. Although ionic liquids are one of the most promising solvents and reaction media, the breakthrough of their catalysis in aerobic oxidation is very challenging due to the difficulty in the direct activation of molecular oxygen. Herein, a family of novel thermally regulated molybdate-based ionic liquids (Mo-ILs) has been designed and developed for the first time toward molecular oxygen activation for highly efficient tandem oxidative catalysis. Three diverse one-pot oxidative cascade processes for the syntheses of various flavones, imines, and benzyl benzoates were achieved with good to excellent yields using the Mo-IL [Bmim]2[MoO4] as a catalyst under air conditions. The results of spectroscopic investigations and quantum-chemical calculations further demonstrated that a thermally regulated proton migration between the cation [Bmim] and anion [MoO4] was the key to forming N-heterocyclic carbene and thereby to effortlessly promoting the generation of ˙O2− active species from molecular oxygen, which results in excellent catalytic performance in these three aerobic tandem oxidations. Our work extends the application area of ILs as the sole catalyst to one-pot aerobic oxidative cascade catalysis, which could have pronounced implications in future work.


 Please wait while we load your content...
Please wait while we load your content...