Selective hydrogenolysis of 2-furancarboxylic acid to 5-hydroxyvaleric acid derivatives over supported platinum catalysts†
Abstract
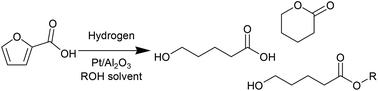
The conversion of 2-furancarboxylic acid (FCA), which is produced by oxidation of furfural, to 5-hydroxyvaleric acid (5-HVA) and its ester/lactone derivatives with H2 was investigated. Monometallic Pt catalysts were effective, and other noble metals were not effective due to the formation of ring-hydrogenation products. Supports and solvents had a small effect on the performance; however, Pt/Al2O3 was the best catalyst and short chain alcohols such as methanol were better solvents. The optimum reaction temperature was about 373 K, and at higher temperature the catalyst was drastically deactivated by deposition of organic materials on the catalyst. The highest yield of target products (5-HVA, δ-valerolactone (DVL), and methyl 5-hydroxyvalerate) was 62%, mainly obtained as methyl 5-hydroxyvalerate (55% yield). The byproducts were mainly ring-hydrogenation compounds (tetrahydrofuran-2-carboxylic acid and its ester) and undetected ones (loss of carbon balance). The catalyst was gradually deactivated during reuses even at a reaction temperature of 373 K; however, the catalytic activity was recovered by calcination at 573 K. The reactions of various related substrates were carried out, and it was found that the O–C bond in the O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C structure (1,2,3-position of the furan ring) is dissociated before C
C structure (1,2,3-position of the furan ring) is dissociated before C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation while the presence and position of the carboxyl group (or methoxy carbonyl group) much affect the reactivity.
C hydrogenation while the presence and position of the carboxyl group (or methoxy carbonyl group) much affect the reactivity.
- This article is part of the themed collection: International Symposium on Green Chemistry 2019


 Please wait while we load your content...
Please wait while we load your content...