Synthesis of water-soluble, fully biobased cellulose levulinate esters through the reaction of cellulose and alpha-angelica lactone in a DBU/CO2/DMSO solvent system†
Abstract
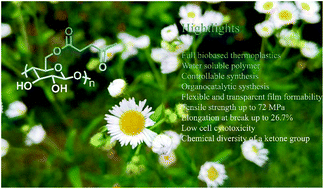
Cellulose esters are important cellulose derivatives with good processing ability and material properties. The design and sustainable synthesis of cellulose esters are prime research topics. Herein, cellulose levulinate esters (CLEs) with degrees of substitution (DSs) ranging from 0.15 to 2.04 were simply prepared through an atom-economic reaction between cellulose dissolved in a newly developed 1.8-diazabicyclo[5.4.0]undec-7-ene/dimethyl sulfoxide/carbon dioxide solvent system and alpha-angelica lactone, a biomass-derived and commercially available platform chemical, without the addition of any external catalysts or condensation reagents. The optimized reaction conditions of 120 °C, 0.5 h, and a 5/1 molar ratio of alpha-angelica lactone to the hydroxyl groups in cellulose resulted in CLEs with appropriate DSs and satisfactory material properties. The structures and thermal properties of the CLEs were characterized by nuclear magnetic resonance spectroscopy, Fourier transform infrared spectroscopy, differential scanning calorimetry, and thermal gravimetric analysis to develop an in-depth understanding of the correlations among the chemical structures, thermal properties, and mechanical properties. A study of solubility in conventional solvents indicated that the CLEs with appropriate DSs were soluble in water, providing a green processing strategy to prepare CLE films with tensile strengths of up to 72 MPa and elongation at break values of up to 26.7%. The morphologies of the films were characterized by scanning electron microscopy and atomic force microscopy. The cytotoxicity of the water-soluble CLEs was also evaluated in vaginal epithelial cells (VK2/E6E7) to identify the potential of the CLEs for biomedical applications.


 Please wait while we load your content...
Please wait while we load your content...