A rigid spirocyclic diol from fructose-based 5-hydroxymethylfurfural: synthesis, life-cycle assessment, and polymerization for renewable polyesters and poly(urethane-urea)s†
Abstract
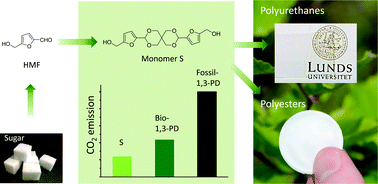
There is currently an intensive development of sugar-based building blocks toward the production of renewable high-performance plastics. In this context, we report on the synthesis of a rigid diol with a spirocyclic structure via a one-step acid-catalyzed acetalation of fructose-sourced 5-hydroxymethylfurfural and pentaerythritol. Preliminary life cycle assessment (LCA) indicated that the spiro-diol produced 46% less CO2 emission than bio-based 1,3-propanediol. Polymerizations of the spiro-diol together with another sugar-based flexible 1,6-hexanediol for the production of polyesters and poly(urethane-urea)s were investigated, and reasonably high molecular weights were achieved when up to 20 and 60 mol% spiro-diol was used for polyesters and poly(urethane-urea)s, respectively. The glass transition temperatures (Tgs) of the polyesters and poly(urethane-urea)s significantly increased upon the incorporation of the rigid spirocyclic structure. On the other hand, it was observed that the spiro-diol was heat-sensitive, which could cause coloration and partial crosslinking when >10% (with respect to dicarboxylate) was used for the polyester synthesis at high temperatures. The results indicated that the polymerization conditions have to be carefully controlled under these conditions. However, when the spiro-diol was used for the synthesis of polyurethanes at lower temperature, the side reactions were insignificant. This suggests that the new spiro-diol can be potentially suitable toward the production of sustainable rigid polyurethane materials like coatings or foams, as well as renewable polyesters after further optimization of the polymerization conditions.


 Please wait while we load your content...
Please wait while we load your content...