Fermentation of pigment-extracted microalgal residue using yeast cell-surface display: direct high-density ethanol production with competitive life cycle impacts†
Abstract
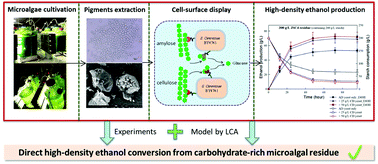
Microalgae have attracted increasing attention as a potential feedstock for biofuel production. However, direct high-density ethanol production from microalgae is not commercially feasible due to the requirement for complex pre-treatments and insufficient enzymatic hydrolysis. In this study, we successfully developed a consolidated bioprocessing (CBP) system using recombinant Saccharomyces cerevisiae displaying synergistic cellulases/amylases on cell surfaces to overcome energy-conversion limitations. As Chlamydomonas sp. JSC4 can accumulate considerable amounts of carbohydrates and pigments (i.e., lutein), performing four rounds of pigment extraction from wet microalgal biomass using acetone was found to significantly eliminate the need for biomass pre-treatment and increase commercial viability. The pigment-extracted JSC4 residues increased ethanol production by 10.7% and 31.6% compared to raw starch and whole JSC4 cells, respectively. The theoretical ethanol production mass and yield from 300 g L−1 of JSC4 material were 73 g L−1 and 64%, respectively, after fermentation for 72 h in the presence of amylase- and cellulase-displaying yeasts, which are dramatically higher than those reported previously. Life cycle assessment (LCA) further revealed that this CBP system has 2.7- to 10.7-fold lower total environmental impact compared to alternative ethanol production methods using microalgal biomass. 2.43 kg ethanol and additional products of 5 g lutein from 1 kg microalgal biomass significantly increased the total economic output to $60.875. Overall, this study successfully demonstrates a feasible cell-surface display fermentation system for use in direct high-density ethanol production from pigment-extracted microalgal material.


 Please wait while we load your content...
Please wait while we load your content...