Conjugated tri-nuclear salen-Co complexes for the copolymerization of epoxides/CO2: cocatalyst-free catalysis†
Abstract
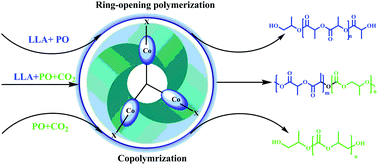
A novel trinuclear salen-Co(III) complex 1 was designed for carbon dioxide (CO2) and propylene oxide (PO) copolymerization. Due to its conjugated structure, the trinuclear complex 1 system showed a synergistic effect with high activity at low catalyst loadings. In contrast, the corresponding mononuclear complex 2 system almost lost its activity under the same conditions. Unexpectedly, a conjugated effect in the absence of a nucleophilic cocatalyst was discovered, in which complex 1 showed unusual activity even under 0.1 MPa pressure of CO2. This is the first example of PO/CO2 copolymerization using salen-Co(III) as the catalyst without the aid of a cocatalyst as the stabilizing cation. Moreover, complex 1 showed great tolerance to H2O in the reaction system, and the activity increased efficiently with the addition of H2O. The induction period was scarcely observed in the copolymerization by in situ spectroscopy. The copolymerization mechanism for complex 1 without any cocatalyst was postulated based on rationally designed experiments, and the pathway provided new insight into the nature of the true initiating intermediate. Complex 1 could also catalyze the copolymerization of lactide (LA), PO and CO2 without a nucleophilic cocatalyst to produce multiblock copolymers. The stabilization and distinct catalytic activity of trinuclear complex 1 could be attributed to the change of the electrophilicity of the Co(III) center generated from the specific electronic interactions among the three salen-Co(III) subunits.


 Please wait while we load your content...
Please wait while we load your content...