Cleavage of lignin C–O bonds over a heterogeneous rhenium catalyst through hydrogen transfer reactions†
Abstract
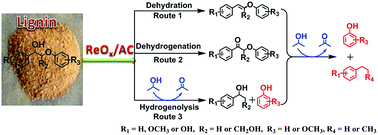
Hydrogenolysis is one of the most popular strategies applied in the depolymerization of lignin for the production of aromatic chemicals. Currently, this strategy is mainly conducted under high hydrogen pressure, which can pose safety risks and is not sustainable and economical. Herein, we reported that heterogeneous rhenium oxide supported on active carbon (ReOx/AC) exhibited excellent activity in the selective cleavage of lignin C–O bonds in isopropanol. High yields of monophenols (up to 99.0%) from various lignin model compounds and aromatic liquid oils (>50%) from lignin feedstock were obtained under mild conditions in the absence of H2. The characterization of the catalyst by X-ray absorption fine structure, X-ray photoelectron spectroscopy and H2-temperature-programed reduction suggested that the activity of ReOx/AC could be attributed to the presence of ReIV–VI. The interaction between the surface oxygen groups of the active carbon and rhenium oxide could also play an important role in the cleavage of the C–O bonds. Notably, an ReOx/AC-catalyzed C–O bond cleavage pathway beyond a typical deoxydehydration mechanism was disclosed. More importantly, 2D-HSQC-NMR and GPC characterizations showed that ReOx/AC exhibited high activity not only in β-O-4 cleavage, but also in the deconstruction of more resistant β-5 and β–β linkages in lignin without destroying the aromatic ring. This study paves the way for the development of rhenium-based catalysts for the controlled reductive valorization of realistic lignin materials through a hydrogen transfer pathway.


 Please wait while we load your content...
Please wait while we load your content...