Recent advances in hydrodeoxygenation of biomass-derived oxygenates over heterogeneous catalysts
Abstract
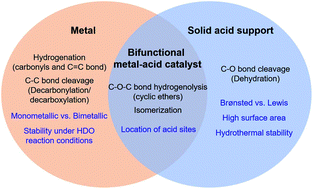
Hydrodeoxygenation (HDO) using heterogeneous catalysts has received considerable attention as a way of converting biomass-derived oxygenates into renewable fuels and chemicals. HDO involves a combination of different reactions such as hydrogenation, hydrogenolysis, decarbonylation, and dehydration. These reactions occur at different catalytic sites, typically with heterogeneous catalysts (e.g., metal, acid, and bifunctional sites), making HDO reactions complex. Therefore, the selection of active site type is critical when designing effective heterogeneous catalysts for HDO processes. Catalyst stability is a major issue in designing HDO catalysts. Traditional catalysts are not stable under HDO conditions (i.e., high partial pressure of water). Metal particles are leached and/or sintered. A solid acid support suffers from phase transformation and losses in surface area. The condensation or polymerization of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C or C
C or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond within biomass and its derived oxygenates is easy to undertake under high temperatures in the HDO process, causing carbon deposition on the catalyst. Poor catalyst stability is a major challenge that needs to be overcome for innovation in HDO technologies. Therefore, herein, we focus on providing an insight into how to design effective catalysts for use in HDO reactions. First, the roles of different catalytic sites in HDO and strategies to stabilize these active sites are discussed. Current achievements in the HDO of different biomass feedstocks (e.g., model compounds and bio-oils) over various heterogeneous catalysts are highlighted. Recent developments in electrochemical HDO processes, which do not require gaseous hydrogen, are discussed. Finally, points to be considered for further research into HDO over heterogeneous catalysts and pending challenges are discussed.
O bond within biomass and its derived oxygenates is easy to undertake under high temperatures in the HDO process, causing carbon deposition on the catalyst. Poor catalyst stability is a major challenge that needs to be overcome for innovation in HDO technologies. Therefore, herein, we focus on providing an insight into how to design effective catalysts for use in HDO reactions. First, the roles of different catalytic sites in HDO and strategies to stabilize these active sites are discussed. Current achievements in the HDO of different biomass feedstocks (e.g., model compounds and bio-oils) over various heterogeneous catalysts are highlighted. Recent developments in electrochemical HDO processes, which do not require gaseous hydrogen, are discussed. Finally, points to be considered for further research into HDO over heterogeneous catalysts and pending challenges are discussed.


 Please wait while we load your content...
Please wait while we load your content...