A strategy for the dissolution and separation of rare earth oxides by novel Brønsted acidic deep eutectic solvents†
Abstract
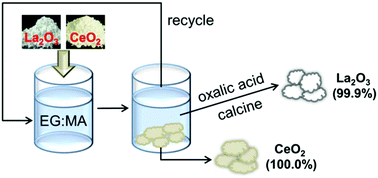
The dissolution and separation of rare earth oxides (REOs) by environmentally benign solvents are important because of the vast high-tech applications of rare earth materials, but they still remain a challenge. In this study, a family of novel Brønsted acidic deep eutectic solvents (DESs) based on carboxylic acids and polyols was designed and prepared. Then, comprehensive characterization and intermolecular interaction studies were carried out on these DESs. Moreover, the dissolution of REOs was achieved by taking advantage of the moderate viscosity and strong acidity of the designed DESs. The optimization experiments showed that ethylene glycol : maleic acid (EG : MA) at the molar ratio of 4 : 1 exhibited considerable solvation ability for the studied REOs. The solubilities of Eu2O3 and La2O3 could reach as high as 0.168 g g−1 and 0.147 g g−1, respectively. The FTIR results showed that the dissolution was caused by H+ released from EG : MA. Oxalic acid was applied to strip the dissolved rare earths and then calcined in order to form the oxides. Moreover, REOs could be separated due to their significant solubility differences. For example, La2O3 and CeO2 were successfully separated; La2O3 was obtained with purity of 99.9%, while Y2O3 and Sm2O3 could be enriched in the precipitation phase (with the purities of 99.97% and 99.94%, respectively). Recycling of the employed DESs for reuse in rare earth separation was also demonstrated. The reported separation process illustrates the potential of the Brønsted acidic deep eutectic solvents for dissolving and processing REOs.


 Please wait while we load your content...
Please wait while we load your content...