Multi-component syntheses of diverse 5-fluoroalkyl-1,2,3-triazoles facilitated by air oxidation and copper catalysis†
Abstract
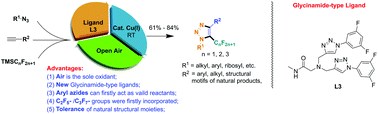
Air is an ideal oxidant due to its abundance, low cost and lack of toxic by-products. However, the catalytic aerobic oxidation reaction using air as an oxidant is still challenging due to its reduced activity and poor selectivity. In this study, a new class of glycinamide-type ligands was successfully synthesized and utilized for the aerobic oxidative reaction via in situ trapping of the formed triazolides with the nucleophilic fluoroalkyl reagents. By this designed oxidative procedure, various 5-fluoroalkyl-1,2,3-triazoles could be synthesized from available fluoroalkyl reagents, terminal alkynes and organic azides via one-pot reactions. Air was used as the sole oxidant; this significantly reduced the cost of this transformation, and the resulting operations and work-up procedures were significantly simplified. Alkyl azides and alkynes bearing various functional groups and natural structural motifs, and even aryl azides, which were ineffective in other reported methods, reacted efficiently under the current conditions. Different fluoroalkyl reagents, including trifluoromethyl trimethylsilane (CF3TMS), pentafluoroethyl trimethylsilane (C2F5TMS) and heptafluoropropyl trimethylsilane (C3F7TMS), could be used effectively for these reactions. The mechanistic study further indicates that the new N,O-ligand can stabilize the Cu(I) catalyst in a multiple binding mode and effectively promote the generation of oxidative coupling products; high efficiency together with a wider substrate scope has enlisted this copper-catalyzed aerobic coupling reaction among the most efficient methods for the preparation of 5-fluoroalkyl 1,2,3-triazoles.


 Please wait while we load your content...
Please wait while we load your content...