Transesterification of soybean oil using a switchable-hydrophilicity solvent, 2-(dibutylamino)ethanol†
Abstract
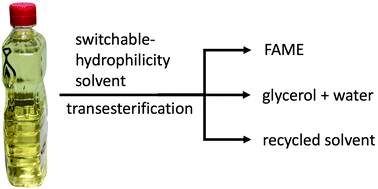
Renewable and environmentally friendly biodiesel has been studied extensively as a replacement for petroleum-derived diesel fuel. Biodiesel contains long chain fatty acid methyl esters (FAMEs) prepared from triglycerides and methanol with basic catalysts such as sodium or potassium hydroxides. The recovery of the basic catalysts is plagued by high water solubility and unwanted soap formation, such that neutralization of the bases with strong acid, rather than recovery, is usually practiced. As an alternative base, switchable-hydrophilicity solvents (SHS) are proposed because they can be readily recovered and reused thanks to their ability to reversibly switch from being hydrophobic to hydrophilic under atmospheric CO2. This novel approach has been tested with 2-(dibutylamino)ethanol (2-DBAE), an SHS, as both solvent and recoverable base catalyst for the transesterification of soybean oil. Carbonated water extracts the SHS from the product FAMEs, while removal of the CO2 from the aqueous stream triggers the separation of the SHS from the water. The FAME yield was approximately 80–85% and the recovery of 2-DBAE was roughly 92%.


 Please wait while we load your content...
Please wait while we load your content...