A rational design of a Pd-based catalyst with a metal–metal oxide interface influencing molecular oxygen in the aerobic oxidation of alcohols†
Abstract
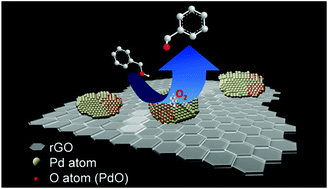
In a green process for selective oxidation of alcohols, the utilization of molecular oxygen as a primary oxidant is the most critical step. Although many palladium (Pd)-based catalysts have shown potential, the role of different Pd-species in the aerobic oxidation reaction is still a matter of discussion. There have been diverse reports, which describe either Pd0 or Pd2+ as the individual species responsible for the aerobic oxidation of alcohols. Herein we demonstrate that the presence of both Pd0 and Pd2+ species with a Pd–PdO interface stabilized on the surface of reduced graphene oxide (rGO) is important for the oxidation of alcohols. With an optimum Pd2+/Pd0 ratio, the catalyst catalyzes the oxidation of benzyl alcohol in water with oxygen, resulting in a turnover frequency (TOF) of up to 18 000 h−1 with 98% selectivity towards the aldehyde. It is proposed that both metallic Pd and its oxide domains, when co-existing with a phase boundary between them, promote the activation of oxygen. On the other hand, rGO provides surface functionalities for the formation and stabilization of Pd–PdO nanoclusters enabling the catalyst to be both stable and reusable. Using histidine as a scavenger for singlet oxygen, we have also determined the importance of oxygen-activation in the reaction. Furthermore, the catalyst is capable of converting various other alcohols into the corresponding carbonyl compounds. Comparison of various catalysts shows that the Pd–PdO@rGO catalyst is the most efficient in terms of TOF, conversion and selectivity for the oxidation of benzyl alcohol using oxygen compared to the reported Pd-based catalysts, particularly when performed under milder reaction conditions. Therefore, the result on Pd-catalyst designing is believed to be of significance for the further developments in the environmentally benign oxidation processes involving molecular oxygen as the oxidant.


 Please wait while we load your content...
Please wait while we load your content...