Glycation by saccharides of different molecular sizes affected the allergenicity of shrimp tropomyosin via epitope loss and the generation of advanced glycation end products
Abstract
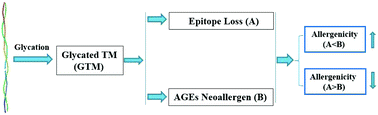
Tropomyosin is the most potent allergen of shrimp that can cause severe food allergy. However, to date, an effective approach to eliminate this allergenicity is still lacking. Glycation is a promising approach that can reduce the allergenicity of shrimp tropomyosin by destroying the epitopes; however, advanced glycation end products (AGEs) are also generated during glycation, which can function as neoallergens to strengthen the allergenicity; therefore, it is hard to tell how the glycation of an allergen with different saccharides affects the allergenicity via epitope loss and neoallergen generation. This study was aimed at the elucidation of how the glycation of tropomyosin (TM) with saccharides of different molecular sizes (glucose, maltose, and maltotriose) affected the allergenicity through epitope loss and the generation of neoallergns that belonged to advanced glycation end products (AGEs). Saccharides of higher molecular sizes (maltotriose) could lead to higher glycated TM than saccharides of smaller molecular sizes (glucose and maltose). Compared with TM, the TM glycated by glucose (TM-G) and maltotriose (TM-MTS) had lower allergenicity and contributed to weaker mouse allergy symptoms; on the other hand, the TM glycated by maltose (TM-M) had no significant impact on the allergenicity due to the generation of AGE-related neoallergens, which might offset the glycation-induced epitope loss. The glycation of TM by maltotriose led to lower generation of AGE neoallergens (e.g. CML) than that in the cases of glucose and maltose; therefore, maltotriose could be applied to desensitize TM-induced food allergy through glycation, and this could be a potential immunotherapy for shrimp allergy patients.


 Please wait while we load your content...
Please wait while we load your content...