α-Bisabolol protects against β-adrenergic agonist-induced myocardial infarction in rats by attenuating inflammation, lysosomal dysfunction, NLRP3 inflammasome activation and modulating autophagic flux
Abstract
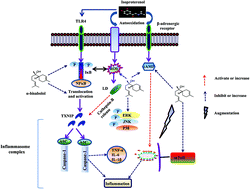
Emerging evidence demonstrates that NLRP3 inflammasome activation, lysosomal dysfunction, and impaired autophagic flux play a crucial role in the pathophysiology of myocardial infarction (MI). Therapeutic strategies targeting NLRP3 activation, lysosomal enzyme release and correcting autophagy have shown beneficial effects in suppressing early inflammatory responses in cardiovascular diseases. Thus, the agents, which inhibit NLRP3 activation and correct lysosome dysfunction and impaired autophagic flux, can be potential drugs for MI. The present study evaluated the effects and elucidated the NLRP3 inflammasome mediated mechanism of α-bisabolol, a dietary sesquiterpene alcohol in a rat model of isoproterenol (ISO)-induced MI. In the present study, male albino Wistar rats were pre- and co-treated with intraperitoneal injection of α-bisabolol (25 mg kg−1) daily for 10 days along with the subcutaneous injection of ISO (85 mg kg−1) at an interval of 24 h for two days (9th and 10th day). ISO injections induced MI as evidenced by the elevated cardiac marker enzyme in serum and altered oxidative stress markers in the total heart and lysosomal fractions. ISO also caused activation of NLRP3 inflammasomes mediating TLR4-NFκB/MAPK signaling pathways and lysosomal dysfunction along with induction and release of proinflammatory cytokines. Interestingly, treatment with α-bisabolol favorably corrected the morphological, histopathological, ultrastructural, biochemical and molecular abnormalities induced by ISO-induced MI in rats. Furthermore, the ultrastructural studies also confirmed the improvement in the autophagic mechanism. The findings of the present study clearly demonstrate that α-bisabolol attenuates oxidative stress and inflammation by inhibiting NLRP3 inflammasome activation and TLR4-NFκB/MAPK signaling pathways along with correcting lysosomal dysfunction and impaired autophagic flux. The underlying pharmacological and molecular mechanism of cardioprotection was attributed to its antioxidant, free radical scavenging and anti-inflammatory properties.


 Please wait while we load your content...
Please wait while we load your content...