Direct experimental evaluation of ligand-induced backbonding in nickel metallacyclic complexes†
Abstract
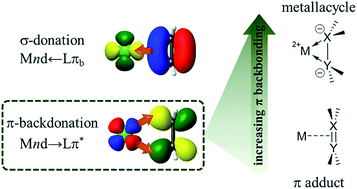
The details of ligand-induced backbonding in nickel diphosphine π complexes are explored using nickel L-edge (3d←2p) X-ray absorption spectroscopy as a means of quantifying the degree of backbonding derived from direct Ni 3d donation into the π ligand. It is observed that backbonding into weakly π acidic ligands such as alkenes and arenes is dominated by contributions from the diphosphine ligand via σ-donation, leading to activated metallacycles with a Ni(0) d10 metal centre. With more strongly π acidic ligands, however, metal contributions to backbonding increase substantially leading to a more electron-deficient metal centre that is best described as having a Ni(I) spectroscopic oxidation state.
- This article is part of the themed collection: Mechanistic processes in organometallic chemistry


 Please wait while we load your content...
Please wait while we load your content...