Multivalent binding of concanavalin A on variable-density mannoside microarrays
Abstract
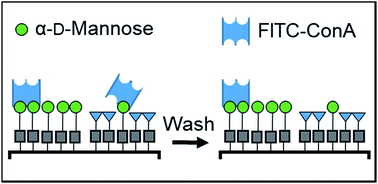
Interactions between cell surface glycans and glycan binding proteins (GBPs) have a central role in the immune response, pathogen–host recognition, cell–cell communication, and a myriad other biological processes. Because of the weak association between GBPs and glycans in solution, multivalent and cooperative interactions in the dense glycocalyx have an outsized role in directing binding affinity and selectivity. However, a major challenge in glycobiology is that few experimental approaches exist for examining and understanding quantitatively how glycan density affects avidity with GBPs, and there is a need for new tools that can fabricate glycan arrays with the ability to vary their density controllably and systematically in each feature. Here, we use thiol–ene reactions to fabricate glycan arrays using a recently developed photochemical printer that leverages a digital micromirror device and microfluidics to create multiplexed patterns of immobilized mannosides, where the density of mannosides in each feature was varied by dilution with an inert spacer allyl alcohol. The association between these immobilized glycans and FITC-labeled concanavalin A (ConA) – a tetrameric GBP that binds to mannosides multivalently – was measured by fluorescence microscopy. We observed that the fluorescence decreased nonlinearly with increasing spacer concentration in the features, and we present a model that relates the average mannoside–mannoside spacing to the abrupt drop-off in ConA binding. Applying these recent advances in microscale photolithography to the challenge of mimicking the architecture of the glycocalyx could lead to a rapid understanding of how information is trafficked on the cell surface.
- This article is part of the themed collection: Nanolithography of biointerfaces


 Please wait while we load your content...
Please wait while we load your content...