Bio-inspired protonic memristor devices based on metal complexes with proton-coupled electron transfer†
Abstract
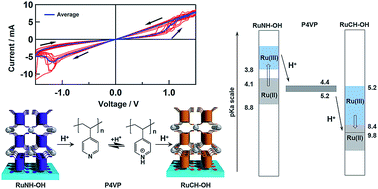
A new type of memristor inspired by bio-membranes is presented, based on the proton movement resulting from proton-coupled electron transfer (PCET) processes in dinuclear Ru complexes, whereby a two-terminal device based on said Ru complexes and a proton-conducting polymer was constructed as a proof-of-concept. Two ITO electrodes were modified separately with dinuclear Ru complexes that bear tetraphosphonic acid linkers at both ends and a 2,6,2′,6′-tetrakis(benzimidazol-2-yl)-4,4′-bipyridine (RuNH-OH) or 1,3,1′,3′-tetrakis(benzimidazol-2-yl)-5,5′-biphenyl (RuCH-OH) bridging ligand, and both ITO electrodes exhibit PCET processes with different Ru(II/III) redox potentials and pKa values. Poly(4-vinylpyridine) (P4VP; pKa = 4–5), a proton-conducting polymer, was sandwiched between the two modified ITO electrodes to construct a two-terminal device of the type ITO|(RuNH-OH)3|P4VP|(RuCH-OH)3|ITO. Initially, the oxidation state of the metal centers in RuNH-OH and RuCH-OH is Ru(II) and Ru(III), respectively. Upon applying a bias voltage between the two ITO electrodes, the high and low current states switch at approximately ±1.10 V due to Ru(II/III) redox reactions. At the RuNH-OH|P4VP and RuCH-OH|P4VP interfaces, a proton is released from Ru(II)NH-OH and subsequently captured by Ru(III)CH-OH through the hydrogen-bonding interaction with the P4VP polymer, which is driven by the changes in the pKa values of the Ru complexes from 4.1–8.8 [Ru(II)NH-OH] to <3.8 [Ru(III)NH-OH] and from <8.4 [Ru(II)CH-OH] to 5.2–9.8 [Ru(III)CH-OH] under these conditions. The redox reactions on the modified Ru films create a large proton gradient between the two electrodes, enhancing the proton conductivity through the P4VP layer (pKa = 4–5). When the applied bias potential was inverted, the pKa gradient returned to the original state and the current decreased. Such a proton-conductivity enhancement is relevant to the transport of protons by proton gradients in bio-membranes. Therefore, the present protonic coordination-network films containing metal complexes that exhibit PCET should open new avenues for the design of a new type of memristor devices mimicking the function of synapses.
- This article is part of the themed collection: New memory paradigms: memristive phenomena and neuromorphic applications


 Please wait while we load your content...
Please wait while we load your content...