Enhanced removal of antibiotics in hospital wastewater by Fe–ZnO activated persulfate oxidation†
Abstract
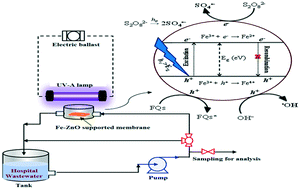
Removal of two widely used antibiotics, flumequine (FLU) and ciprofloxacin (CIP), in synthetic and real hospital wastewaters was investigated using Fe-doped zinc oxide (Fe–ZnO) and UV-A irradiation. Fe–ZnO was supported on a cellulose acetate membrane and integrated in a recirculation reactor system, designed for continuous oxidative treatment of contaminated wastewaters. Addition of a low concentration of persulfate (e.g. 0.5 mM) improved considerably the degradation extent, where sulfate radicals were found to be the most involved oxidizing species. Oxidation rate constants decreased in mixture systems with respect to those of individual compounds, because of competition effects with radical species. Wastewater constituents like those found in hospital effluents affected the removal of both CIP and FLU, but CIP degradation is less affected probably because of its higher reactivity with sulfate radicals. Inorganic anions altered slightly the oxidation rate, while the organic compounds commonly found in wastewaters considerably reduced the degradation efficiency. ZnO doped with Fe exhibited photoactivity higher than bare zinc oxide and commercial TiO2, especially in hospital wastewater. The Fe–ZnO supported membrane can also be re-used for several oxidation runs, making this system quite suitable for larger applications in wastewater treatment processes.


 Please wait while we load your content...
Please wait while we load your content...