Effects of pH on AHL signal release and properties of ANAMMOX granules with different biomass densities†
Abstract
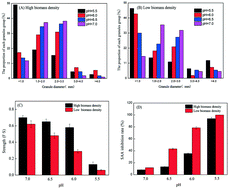
This study investigates the effects of pH on the stability and activity of ANAMMOX granules with different biomass densities from the perspective of N-acyl-homoserine lactone (AHL)-based quorum sensing (QS). Results clearly showed that the effects of pH on the release of AHLs in ANAMMOX granules and the properties of the granules are closely related to biomass density under neutral and weakly acidic conditions (pH 6.0–7.0). Under these conditions, pH had greater adverse effects on the granules with low biomass density (L-granules) than the granules with high biomass density (H-granules). When pH decreased from 7.0 to 6.0, the H-granules were able to release abundant AHLs (the release amounts of C6-HSL and C8-HSL reached up to 1482.4 and 1194.7 ng L−1, respectively, at pH 6.0), which further promoted extracellular polymeric substance (EPS) secretion, serving to maintain the stability and activity of the granules under neutral and weakly acidic conditions. But, L-granules could not release enough AHLs, leading to the low EPS content and deterioration in the stability and activity of the granules under these conditions. However, under neutral and weakly acidic pH conditions, the generation mechanism of AHLs in L-granules could be activated by short-term (10 d) exogenous addition of C6-HSL and C8-HSL, causing excessive endogenous release of C6-HSL and C8-HSL, which was conducive to improving the stability and activity of L-granules under adverse weakly acidic pH conditions. Under peracid (pH 5.5) and alkaline (pH 7.5–9.0) conditions, the effects of pH on AHL release and granule characteristics were not related to biomass density. At pH 5.5, the QS system of the granules was destroyed, and the release amounts of AHLs in H-granules and L-granules were very low, leading to poor stability and low activity of the granules. Under alkaline conditions (pH 7.5–9.0), the release amounts of AHLs in H-granules and L-granules were significantly reduced with the increase in pH, and their stability and activity also decreased significantly, which was related to the degradation of AHLs under alkaline conditions.


 Please wait while we load your content...
Please wait while we load your content...