Influence of mineral water constituents, organic matter and water matrices on the performance of the H2O2/IO4−-advanced oxidation process
Abstract
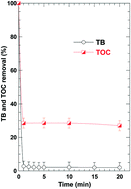
We have recently reported that the reaction of H2O2/IO4− could be a new advanced oxidation process (AOP) for water treatment [N. E. Chadi, S. Merouani, O. Hamdaoui, M. Bouhelassa and M. Ashokkumar, H2O2/periodate (IO4−): a novel advanced oxidation technology for the degradation of refractory organic pollutants, Environ. Sci.: Water Res. Technol., 2019, 5, 1113–1123]. Experiments were conducted in deionized water using toluidine blue (TB) as a model pollutant, in which the impact of all operating conditions was clarified. Herein, we report, for the first time, the impact of mineral water constituents (salts), different kinds of organic additives (surfactants, sucrose, glucose and carboxylic acids) and water matrices (natural mineral water, seawater and treated wastewater effluent) on the performance of this emerging AOP. It was observed that the presence of salts decreased the degradation efficiency of the process. The one exception is that of bicarbonate which notably accelerated the TB degradation rate but only at low HCO3− concentration. Similarly, the presence of organic competitors diminished the efficiency of TB degradation through consuming radicals and oxidizing species generated by the reaction of H2O2 with periodate. The quenching impact of organic competitors and salts increased with their concentrations in the solution. Degradation trials conducted in real environmental matrices showed that more than 50% of the process efficacy was lost when the pH of the matrices was adjusted to 5.4, but a lower loss was observed when the process was operated at natural pH (∼7.6). All these findings were discussed and interpreted with regard to the reactivity of radicals formed with additives present in the system.


 Please wait while we load your content...
Please wait while we load your content...