A novel strategy using peroxymonosulfate to control the formation of iodinated aromatic products in treatment of phenolic compounds by permanganate†
Abstract
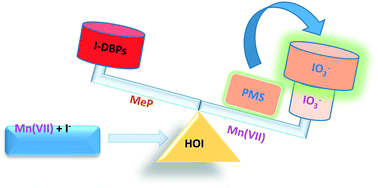
Iodinated aromatic products of high toxicity can be generated during permanganate (Mn(VII)) oxidation of phenolic compounds in the presence of iodide (I−), mainly due to the interactions between phenols and hypoiodous acid (HOI) formed in situ from Mn(VII) oxidation of I−. In this work, the feasibility of peroxymonosulfate (PMS) addition to control the formation of iodinated aromatic products during Mn(VII) oxidation of a representative phenolic compound methylparaben (MeP, a widely used preservative) in the presence of I− under various conditions was investigated. The HOI-enhanced transformation of MeP by Mn(VII) was generally inhibited by the addition of PMS, and stronger inhibitive effect at higher PMS concentration or higher pH was obtained resulting mainly from the competitive oxidation of HOI to iodate (IO3−) by PMS vs. MeP. Formation of iodinated aromatic products from MeP treated by Mn(VII) in the presence of I− (i.e., mono- and di- iodinated MeP) at pH 5, 7, and 9 in synthetic water as well as authentic water was also decreased by the addition of PMS, especially at higher PMS concentration, and IO3− formation was promoted in parallel under the same conditions. A similar inhibitory effect of PMS on the formation of iodinated aromatic products from MeP by manganese dioxide (MnO2) in the presence of I− was also observed. These results suggest that PMS may be a promising oxidant for controlling iodinated aromatic products during manganese oxidation processes.


 Please wait while we load your content...
Please wait while we load your content...