Membrane-separated electrochemical latrine wastewater treatment†
Abstract
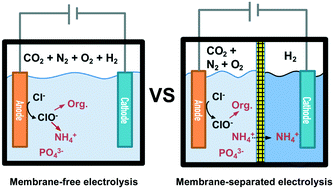
Electrolysis is demonstrated to be useful for onsite latrine wastewater treatment. Improved wastewater treatment efficiencies are achieved through the use of a combination of cation and anion exchange membranes (CEM/AEM) in electrochemical reactors. Compared to a membrane-free electrolysis cell, the separation of anodic and cathodic chambers using a CEM separator is shown to reduce energy consumption by 51% for COD removal and 87% for NH4+ removal. Furthermore, 51% of the initial [NH4+]0 is recovered via electrodialysis. CEM-separated electrolysis is shown to produce 39% less ClO3− and 92% less chlorinated organic by-products than in a membrane-free reactor. Helminth (Ascaris suum) eggs, which yield parasitic worms, are very resistant to conventional disinfection methods. Membrane-free electrolysis only inactivates 15% of the dosed helminth eggs (100 eggs per mL). In CEM-separated electrolysis, a combination of low pH and in situ chlorine production in the anodic chamber results in 85% inactivation of helminth eggs. In addition, H2, which is produced in the cathodic chamber of the CEM-separated reactor, is directly converted to electricity using a hydrogen–air fuel cell. The hydrogen energy produced during electrolysis is estimated to reduce the overall energy cost of operation by 20%. Recovery of 85% of the initial [PO43−]0 and pH neutralization are achieved by treating the acidic effluent of a CEM-separated electrolysis cell in an AEM-separated electrolysis cell. A one-month continuous operation demonstrates the potential of using both the CEM and AEM during electrolysis to achieve more efficient wastewater treatment while, at the same time, recovering NH4+ and PO43−, for eventual use as agricultural fertilizers.
- This article is part of the themed collections: Best Papers of 2019 from RSC’s Environmental Science family journals and Best Papers 2019 – Environmental Science: Water Research & Technology


 Please wait while we load your content...
Please wait while we load your content...