Humic acid regulation of the environmental behavior and phytotoxicity of silver nanoparticles to Lemna minor†
Abstract
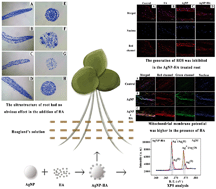
The environmental transformations of silver nanoparticles (AgNPs) are correlated with their behaviors and ecological risks. Humic acid (HA) is ubiquitous in natural water environments. However, the influence of HA on the environmental behavior, bioavailability, and toxicity of AgNPs is little known. This study investigated the processes of AgNP bioaccumulation in and toxicity to Lemna minor in an aquatic environment. AgNP effects were assessed in the presence of HA, which has been shown to interact with AgNP and modify its behavior and toxicity to organisms. Results showed that AgNP dissolution declined with the addition of HA. Carboxylate groups and C–O and C–O–C bonds were found to be involved in the HA and AgNP interaction, while XPS results suggested that HA caused Ag+ to be reduced to Ag(0). The addition of HA inhibited the absorption and accumulation of Ag in Lemna minor roots and leaves. When exposed to AgNP treatments alone, the plant morphology and ultrastructure were damaged by root atrophy and a reduction in vessel number and cross-sectional areas (CSAs) was observed. However, the addition of HA mitigated AgNP toxicity, resulting in no significant difference in the chlorophyll biomass and concentration between the AgNP–HA and unexposed treatment groups. The addition of HA was found to inhibit the generation of ROS and the depolarization of mitochondrial membrane potential (ΔΨm) caused by AgNPs in root cells. Overall, these results establish the mechanism of the effect of HA on AgNP behavior and highlight the influence of HA on the uptake and toxicity of AgNPs to Lemna minor.


 Please wait while we load your content...
Please wait while we load your content...