Efficient removal of low-concentration organoarsenic by Zr-based metal–organic frameworks: cooperation of defects and hydrogen bonds†
Abstract
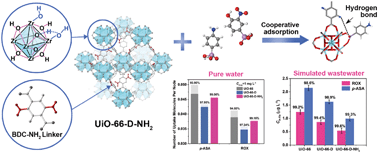
As an emerging micropollutant, organoarsenic has caused global ecosystem pollution, due to its degradation to highly toxic inorganic arsenic in the environment. Therefore, to remove the low-concentration organoarsenic from arsenic compounds from water, materials with high adsorption affinity are in urgent need. Herein, we reported an amino-modified metal–organic framework (MOF) with defects (UiO-66-D-NH2) to adsorb two typical organoarsenic forms p-arsanilic acid (p-ASA) and roxarsone (ROX). The adsorption affinity of UiO-66-D-NH2 was 3.8 and 3.0 times higher than that of original UiO-66 and defective UiO-66 without amino modification (UiO-66-D), while the removal rate exceeded 99.1% when the initial concentration of organoarsenic decreased to 1 mg L−1. UiO-66-D-NH2 also showed excellent adsorption behavior in simulated wastewater, with residual arsenic concentrations lower than 0.54 μg L−1. Mechanism studies showed a cooperation of the defects and –NH2 groups in UiO-66-D-NH2. The defects provided more Zr–OH sites in the metal node, leading to transformation of the As–O–Zr coordination from monodentate mononuclear to bidentate binuclear configuration, while –NH2 groups formed hydrogen bonds with both p-ASA and ROX as a secondary interaction to further reinforce the as-formed As–O–Zr complex. This work revealed that making cooperation by defect creation and amino modification could be an effective method to enhance the adsorption affinity of MOFs to low-concentration organoarsenic.


 Please wait while we load your content...
Please wait while we load your content...