Preventing viral disease by ZnONPs through directly deactivating TMV and activating plant immunity in Nicotiana benthamiana†
Abstract
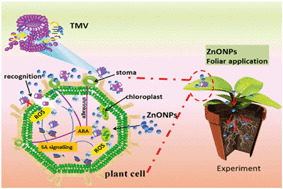
One of the most difficult challenges in crop production is viral infection, which is a tremendous threat to human food security, as an effective control strategy is lacking. Interactions among nanoparticles, plant phytopathogens, and plant responses have attracted increasing attention for antiviral activity; moreover, the influence of nanoparticles on plants supports the idea that nanoparticles can be employed as a new antiviral strategy in agriculture. Zinc oxide nanoparticles (ZnONPs) and silica nanoparticles (SiO2NPs) were fabricated to control Tobacco mosaic virus (TMV) infection in vitro and in vivo. TMV particles exhibited substantial aggregation and breakage in vitro after pretreatment for 2 h with the NPs (ZnONPs or SiO2NPs). These mixtures were then inoculated onto tobacco plants and showed significantly lower virus colonization than the control group 2 days after inoculation. Conversely, 7 days after inoculation, the inactivation effects were insufficient to resist viral replication and accumulation. Significantly, by contrast, daily foliar spraying of NPs onto the leaves of tobacco for 12 days markedly inhibited TMV replication owing to the activation of the plant defense and growth response. Specifically, reactive oxygen species accumulation, peroxidase activity, catalase activity and systemic resistance-related genes (PR1 and PR2) were upregulated in NP-treated plants. Further study showed increases of 162% and 517% in the SA and ABA phytohormone levels induced by ZnONPs. Interestingly, the NPs also increased the dry weight and fresh weight of tobacco plants compared with the control. We also found that NPs can also be absorbed by the leaves and transported throughout the plant, implying a possible synergistic nutritional provision and means of immune regulation. This strategy of developing NPs as new antiviral materials in agriculture broadened the avenues for applying and researching viral materials.


 Please wait while we load your content...
Please wait while we load your content...