Emerging investigator series: linking chemical transformations of silver and silver nanoparticles in the extracellular and intracellular environments to their bio-reactivity†
Abstract
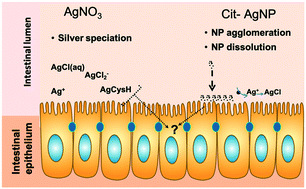
The fish intestine is an important barrier for environmental toxicants, including metals and metal nanoparticles. Tracking chemical transformation at the interface between the intestinal epithelium and the intestinal lumen can inform us about chemicals' bio-reactivity and toxicity but is challenging due to the lack of appropriate models. To allow for such investigations, a model of the fish intestine derived from rainbow trout (Oncorhynchus mykiss), the RTgutGC cell line, was used. Cells were exposed to silver nitrate (AgNO3) or citrate coated silver nanoparticles (cit-AgNPs) in Leibovitz's L-15 medium without amino acids and vitamins (L-15/ex), which allowed the determination of the extracellular silver species using a chemical equilibrium model. X-ray absorption spectroscopy (XAS) was used to track intracellular silver speciation. Cellular toxicity, silver accumulation, and metallothionein (MT) mRNA levels were also measured. Cells accumulated the same concentrations of silver when exposed to equimolar amounts (i.e. 1, 5 and 10 μM) of AgNO3 or cit-AgNPs. However, AgNO3 was shown to be more toxic than cit-AgNPs. Intracellular silver speciation changed over time in both exposure series. After 1 hour, intracellular silver speciation was dominated by chloride complexation in both exposures. After 24 and 72 hours of exposure to cit-AgNPs, ∼7% of silver was complexed to cysteine, whereas the remaining silver was AgNPs. In cells exposed to AgNO3 for 72 hours, 97% of Ag was complexed to cysteine. A significant increase, compared to controls, in metallothionein mRNA levels at 24 and 72 hours of exposure to AgNO3 and cit-AgNPs can explain the formation of Ag–cysteine complexes. In summary, these data show that silver chloride species are bioavailable and that complexation to cysteine scavenges intracellular dissolved silver ions, thus preventing toxicity. Silver nanoparticles present a similar but attenuated toxic response to AgNO3. Thus, at least in acute exposures, existing risk assessment for dissolved silver species could be protective for nanosilver.
- This article is part of the themed collection: Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...
