The effect of water on the heterogeneous reactions of SO2 and NH3 on the surfaces of α-Fe2O3 and γ-Al2O3†
Abstract
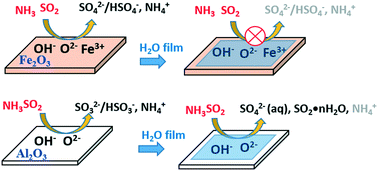
In this study, a wall-coated flow tube reactor, in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) and ion chromatography (IC) were used to investigate the effect of water on the heterogeneous reactions of SO2 and NH3 on the surfaces of α-Fe2O3 and γ-Al2O3. Flow tube results show that water exhibited little effect on the initial uptake coefficients (γBET) of SO2 but the addition of NH3 increased the γBET at various relative humidity (RH) values on both α-Fe2O3 and γ-Al2O3. The γBET of SO2 in the presence of NH3 was negatively RH dependent on α-Fe2O3, which decreased from (8.80 ± 2.30) × 10−6 at 9% RH to (2.24 ± 0.71) × 10−6 at 85% RH. In contrast, the γBET of SO2 in the presence of NH3 on γ-Al2O3 increased from (1.32 ± 0.45) × 10−6 at 10% RH to (2.56 ± 0.39) × 10−6 at 74% RH. Surface product analysis by DRIFTS and IC shows that ammonium sulfate-like species formed, whereas the variation of SO42− and NH4+ with RH differed on Fe2O3 and Al2O3. With the increase of RH, sulfate tended to transform into bisulfate on Fe2O3 with copresence of NH3, whereas surface-coordinated sulfite transformed to water-solvated sulfite and water-solvated sulfate species on Al2O3 regardless of the presence of NH3. During this process, the amount of sulfate decreased with RH on Fe2O3, as opposed to that on Al2O3; yet the NH4+ was inhibited to form by adsorbed water on both surfaces. The different interaction mechanisms among water, NH3, and SO2 on Fe2O3 and Al2O3 as well as the atmospheric implications have been proposed.


 Please wait while we load your content...
Please wait while we load your content...