Understanding the stability of nanoplastics in aqueous environments: effect of ionic strength, temperature, dissolved organic matter, clay, and heavy metals†
Abstract
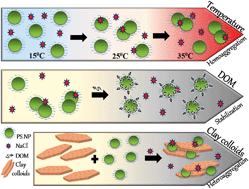
Nanoplastics (NPs) are one of the most dangerous fractions of plastics because of their possible eco-toxicological impacts. NP stability and transport are highly influenced by various environmental factors, which warrants the necessity to understand their fate in ambient water systems. This study investigates the polystyrene (PS) NP stability under the effect of varying ionic strength, temperature, dissolved organic matter (DOM), inorganic soil colloids and heavy metal salts using the dynamic light scattering technique. Controlled studies were used to examine the aggregation of NPs in the presence of natural river water (RW), groundwater (GW), and seawater (SW). Results highlight that, at all studied temperatures, divalent cations had a greater influence on the aggregation rate of NPs as compared to monovalent cations whereas for the same salt, a drop in temperature tended to increase the stability. A rise in critical coagulation concentration (CCC) by 1.6 and 2.4 times for NaCl and CaCl2 was observed, respectively, at 15 °C as compared to 35 °C. Steric repulsion produced by DOM stabilizes NPs shifting the CCC value to a higher salt concentration for NaCl. However, faster aggregation with CaCl2 due to complexation was notable. The clay colloids participate in heteroaggregation with NPs under the influence of salts; this was confirmed using cryo-TEM. Heavy metal salts such as ZnCl2 and CdCl2 had interactions with PS NPs similar to that presented by CaCl2 but showed independent behaviour in the presence of HgCl2, due to metal speciation under different redox conditions. The concentration of salts and organic substances in the complex matrix of natural water results in the least stable NPs in SW > RW > GW. The results of this study contribute to the fundamental understanding of the fate of NPs in complex aquatic environments.


 Please wait while we load your content...
Please wait while we load your content...