Emerging investigator series: connecting concepts of coinage metal stability across length scales†
Abstract
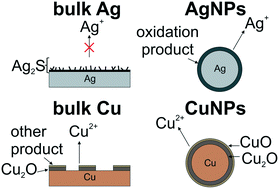
At all length scales, the stability of materials is impacted by their surrounding environment. However, it has now been observed that materials with different dimensions can exhibit markedly different responses to their environment, even for the same material composition. For example, the deterioration of nanomaterials is complicated by the unique chemical and physical properties that arise from changes to their size, shape, and/or surface chemistry. A first step in understanding and predicting nanomaterial stabilities is to leverage the decades of work dedicated to understanding the environment-specific deterioration mechanisms of analogous bulk materials. Then, unique nanoscale properties can be accounted for and used to understand both similarities and differences in deterioration behavior across these length scales. In this review, we specifically consider the stability of group 11 coinage metal surfaces: copper, silver, and gold. We first summarize the chemical mechanisms of environmentally-driven deterioration of these metals in the bulk. We then apply this knowledge to interpret stability studies of nanoparticles made from the same elements. Ultimately, we find that while the reactivity of gold and copper is relatively consistent across length scales, the composition of products formed on silver surfaces differs significantly. These results have important implications for predicting and controlling both desirable and undesirable metal nanoparticle deterioration processes.
- This article is part of the themed collections: Nanocircular Economy Papers 2014-2022, Best Papers 2019 – Environmental Science: Nano and Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...
