Ion specific effects of monovalent cations on deposition kinetics of engineered nanoparticles onto the silica surface in aqueous media†
Abstract
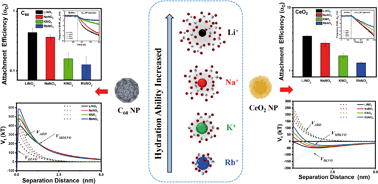
Ion specific effects have been proved to be an important factor controlling the stability and dynamics of engineered colloidal systems; however, there has been limited research on the ion specific effects on the transport behaviors of engineered nanoparticles (ENPs). In the present study, the initial deposition kinetics of fullerene (C60) and ceria (CeO2) NPs in first-group monovalent cationic solutions (i.e., MNO3, M = Li+, Na+, K+ and Rb+) were quantitatively investigated, by employing a laboratory quartz crystal microbalance with the dissipation monitoring (QCM-D) technique. The influence of common model biomacromolecules, including a polysaccharide (alginate) and protein (bovine serum albumin (BSA)), was reported as well. The results demonstrated that the cations can decrease the deposition kinetics of different charged NPs with decreasing hydration degree of additive cations in the order Li+ > Na+ > K+ > Rb+. The variance in the deposition behaviors of NPs at fixed electrolyte concentration can be quantitatively explained by a modified DLVO theory that introduced hydration forces, which induced short range repulsion within the model that dominated the colloidal interactions of NPs with the surface and resulted in these specific effects. Moreover, the two biomacromolecules impacted the deposition of both NPs in an opposite manner: BSA could enhance the attachment of NPs on the silica surface, while alginate can decrease the deposition and enhance their mobility in aqueous solutions, dependent on the molecular complexity. These findings are helpful for understanding the deposition behavior of NPs under the effect of ionic specificity and giving quantitative evidence for the role of counterion hydration in the interactions between electrostatically charged ENPs and the environmental surface in aqueous media.


 Please wait while we load your content...
Please wait while we load your content...