Emerging investigator series: protein adsorption and transformation on catalytic and food-grade TiO2 nanoparticles in the presence of dissolved organic carbon†
Abstract
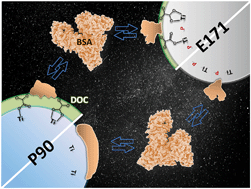
The inherent physicochemical properties of engineered nanomaterials (ENMs) are known to control the sorption of proteins, but knowledge on how the release of ENMs to the environment prior to protein exposure affects this reaction is limited. In this study, time-resolved, in situ infrared spectroscopy was used to investigate the sorption of a model protein, bovine serum albumin (BSA), onto two different types of titanium dioxide (TiO2) ENMs (catalytic-grade P90 and food-grade E171) in the presence and absence of a simple dissolved organic carbon molecule, oxalate. Infrared spectroscopy results showed that oxalate adsorbed to P90 through chemisorption interactions, but it adsorbed to E171 through physisorption interactions due to the presence of inherent surface-bound phosphates. Secondary structure and two-dimensional correlation spectroscopy analyses showed that BSA interacted with and unfolded on the surface of P90, but not E171, presumably due to the repulsive forces from the negatively charged phosphates on E171. When oxalate was pre-adsorbed to either P90 or E171, the unfolding of BSA occurred, but along different pathways. This suggests both the “outer” surface chemistry (e.g., oxalate layers) and the mechanism by which this layer is bound to the ENM play a significant role in the adsorption of proteins. Collectively, the results indicate the exposure of ENMs to natural and engineered environments prior to biological uptake affects the resulting protein corona formation, and thus the transport and bioactivity of ENMs.
- This article is part of the themed collection: Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...
