Transformation of Ag ions into Ag nanoparticle-loaded AgCl microcubes in the plant root zone†
Abstract
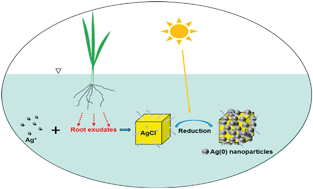
Natural formation of metal nanoparticles is an important pathway that will modify the fate, behavior, and biological availability of heavy metal ions in the environment. Most work has focused on the ability of natural organic matter (NOM) and extracellular polymeric substances (EPS) to convert metal ions into nanoparticles. However, plant roots, ubiquitous in soil and aquatic environments, may have a significant role in the formation of naturally occurring metal nanoparticles. This work demonstrates the importance of plant roots and associated exudates in mediating the transformation of Ag+ in the presence of sunlight. Using Ag+ as the starting material, transformation took place in three steps: 1) formation of AgCl microcubes (μAgCl) through complexation of Ag+ by plant-released chloride ions in root exudates; 2) stabilization of μAgCl by biomolecules in root exudates; 3) partial photoreduction of μAgCl to Ag(0) nanoparticles (nAg) facilitated by exudate biomolecules. Morphological and compositional changes were observed by scanning electron microscopy with energy dispersive X-ray spectroscopy (SEM-EDS) on the particles from 0–24 h: cubic AgCl microcrystals were converted into cauliflower-shaped core–shell structures with nAg clusters as the shell and μAgCl as the core. The quantification of Ag+, μAgCl and nAg species over time demonstrates that the transformation kinetics fit (R2 = 0.99) a second-order reaction (k = 1.11 mM−1 h−1). The discovery of plant root exudate-mediated phototransformation of Ag+ adds new knowledge to our understanding of Ag transformation in the plant root zone and will guide the assessment of both exposure and risk in the environment.


 Please wait while we load your content...
Please wait while we load your content...
