Photochemical interactions between n-Ag2S and n-TiO2 amplify their bacterial stress response†
Abstract
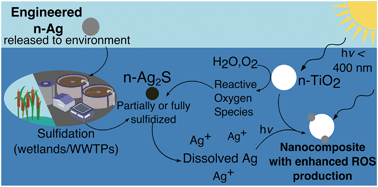
Under the reducing conditions of wetlands and anaerobic wastewater treatment processes, nanosilver (n-Ag) released from consumer products reacts with sulfide to form Ag2S, which is believed to be stable and practically benign. However, in the natural environment, these nanosized silver sulfide particles (n-Ag2S) may interact with light and other nanomaterials to create oxidizing conditions that may destabilize n-Ag2S, releasing Ag+ and causing stress to microorganisms. We investigate this scenario by monitoring the ATP levels of E. coli exposed to mixtures of n-Ag2S and n-TiO2, another nanomaterial with high likelihood of environmental release. Under dark conditions, we find that sulfidation increases the threshold concentration for microbial stress relative to n-Ag. Under simulated solar irradiation (SSI), exposure to n-Ag2S alone does not depress bacterial ATP levels in the concentration range tested (≤1 mg L−1 as Ag). However in the presence of 1 mg L−1 n-TiO2 and under SSI, n-Ag2S causes synergistic toxic stress and enhances the production of reactive oxygen species (ROS). Based on the photochemistry of these nanomaterials and our measurements of dissolved Ag, we propose that under environmental conditions the ROS produced by n-TiO2 dissolves n-Ag2S, releasing Ag+ that can then be photoreduced on the surface of n-TiO2 to form a highly photoactive and phototoxic nanocomposite. This work reveals that under oxidizing conditions n-Ag2S is prone to transformations that cause microbial stress. Overall, we provide further evidence that chemical interactions between multiple nanomaterials under irradiation can dramatically change their toxic effects.


 Please wait while we load your content...
Please wait while we load your content...