Displacement reactions between environmentally and biologically relevant ligands on TiO2 nanoparticles: insights into the aging of nanoparticles in the environment†
Abstract
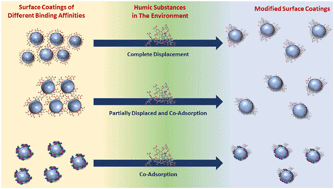
Coatings on nanoparticle (NP) surfaces play a key role in dictating their behavior in the environment. For metal oxide NPs, the physicochemical processes of dissolution, aggregation, and reactivity are all impacted by surface coatings. The current study focuses on adsorption and displacement reactions of different molecular and biological species representative of different coatings on TiO2 NP surfaces. These different species include ascorbic acid (AA), citric acid (CA), humic acid (HA) and bovine serum albumin (BSA) protein. Surface adsorption, desorption, and displacement reactions of these four species on 20 nm TiO2 NPs were investigated using attenuated total reflectance-Fourier transform infrared spectroscopy. Further insights of these reactions and on the behavior of TiO2 with these coatings were gained from quartz crystal microbalance with dissipation measurements, dynamic light scattering and sedimentation studies to investigate nanoparticle stability. Our results show that HA adsorbs strongly onto TiO2 NP surfaces at neutral pH. In contrast, smaller acidic molecules such as AA and CA bind more weakly and, as a result, HA can displace these molecules from the TiO2 surface as determined by two-dimensional correlation spectroscopy. In the case of BSA, HA does not displace the protein but instead co-adsorbs on the nanoparticle surface. Our results show that the relative binding affinity to the surface depends on the ability of different functional groups to interact with the surface and through non-specific surface interactions that become important for species with higher molar mass. To our knowledge, this is the first time that these types of displacement reactions on TiO2 NPs have been investigated and probed with in situ techniques. The insights that this work provide are of relevance in understanding the fate of nanoparticles as ligand displacement reactions will modify the stability of these nanoparticles during their transport in the environment, nanoparticle agglomeration and their interactions with biological systems.


 Please wait while we load your content...
Please wait while we load your content...
