Mechanisms limiting the release of TiO2 nanomaterials during photocatalytic cement alteration: the role of surface charge and porous network morphology†
Abstract
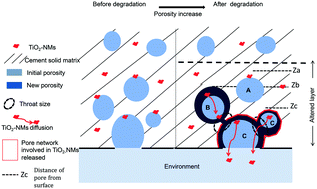
Nanomaterials have been widely used in a wide range of manufactured products in order to improve/add new properties. Photocatalytic cement maintains clean and white wall fronts and also provides interesting air pollution-reducing properties due to the photocatalytic activity of incorporated TiO2-NMs. However, despite these environmental benefits, there is evidence of the release of TiO2-NMs during cement use. Therefore, it is crucial to understand the parameters controlling TiO2-NM release. The zeta potentials of TiO2-NMs were investigated from the unaltered core to the cement altered surface using simulated cement pore waters. The mineralogy and chemical composition of the altered layer were investigated using X-ray diffraction (XRD) and micro X-ray fluorescence spectroscopy (micro-XRF). Finally, the pore network morphology was fully analyzed using X-ray computed tomography at both micro and nano-scales (micro and nano-CT) and quantified using 3D morphological software (i-Morph). This study provides evidence that the TiO2-NM release comes from a very thin “active surface layer” (thickness less than 20 μm) where both the cement surface chemistry and the pore network appear to be favorable for the TiO2-NM diffusion. The pore volume connected to the surface with a throat size of ≥1016 nm appears to control and be used as a predictor for TiO2-NM release.


 Please wait while we load your content...
Please wait while we load your content...