Photolysis of graphene oxide in the presence of nitrate: implications for graphene oxide integrity in water and wastewater treatment†
Abstract
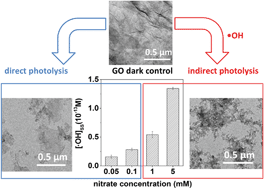
Despite the widespread use of graphene oxide (GO) in diverse applications and increasing interest in its inclusion in some water treatment devices, mechanistic understanding of photochemical GO transformations is limited. This is an important knowledge gap relevant to GO performance and durability. We examined the reaction pathways and products of a GO suspension under UV irradiation in the presence of nitrate, a common anion in water and wastewater treatment processes. As the nitrate concentration increased, the dominant pathway of GO transformation changed from direct photolysis to indirect photolysis enhanced by the production of hydroxyl radicals (˙OH) during UV irradiation of nitrate. At environmentally relevant concentrations (e.g., 1 mM), nitrate induced significant fragmentation of the GO nanostructure. The significant effects of ˙OH on GO morphology and surface properties were verified by negative-control tests, including deoxygenation of the suspension, reactive oxygen species (ROS) inhibition and radical trapping, and by γ-radiolysis, known to generate a single ROS: ˙OH. Supplemental photolysis experiments conducted using graphite demonstrated that the main reaction pathways of the indirect photolysis of GO not only include the oxidation reactions between ˙OH and the oxidized domains of GO, but also the electrophilic addition reaction between ˙OH and the aromatic domains. These findings have significant implications for GO integrity and durability in systems involving incidental or purposeful exposure to UV irradiation.
- This article is part of the themed collection: Research from our newest ESPI Editorial Board members


 Please wait while we load your content...
Please wait while we load your content...