Geochemical conditions conducive for retention of trace elements and radionuclides during shale–fluid interactions†
Abstract
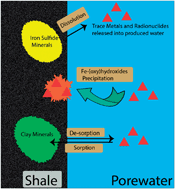
Produced water generated during unconventional oil and gas extractions contains a complex milieu of natural and anthropogenic potentially toxic chemical constituents including arsenic (As), chromium (Cr), and cadmium (Cd), naturally occurring radioactive materials (NORMs) including U and Ra, and a myriad of organic compounds. The human-ecological health risks and challenges associated with the disposal of produced water may be alleviated by understanding geochemical controls on processes responsible for the solubilization of potentially hazardous natural shale constituents to produced water. Here, we investigated, through a series of batch treatments, the leaching behavior of As, Se, Cu, Fe, Ba, Cr, Cd, and radioactive nuclides U, Ra from shale to produced water. Specifically, the effect of four major controls on element mobility was studied: (1) solution pH, (2) ionic strength of the solution, (3) oxic–anoxic conditions, and (4) an additive used in fracking fluid. The mobilization of metals and metalloids from shale was greatest in treatments containing sodium persulfate, an oxidant and a commonly used additive in fracture fluid. In the high ionic strength treatments, dissolved Ba concentrations increased 5-fold compared to low ionic strength treatments. Overall, anoxic conditions superimposed with low pH resulted in the largest increase of dissolved metals and radionuclides such as Ra. Overall, our results suggest that (1) limiting pore water acidification by injection of alkaline fluid in carbonate-low shale and (2) minimizing strong oxidizing conditions in shale formations may result in cost-effective in situ retention of produced water contaminants.


 Please wait while we load your content...
Please wait while we load your content...