Intramolecular electronic coupling in porous iron cobalt (oxy)phosphide nanoboxes enhances the electrocatalytic activity for oxygen evolution†
Abstract
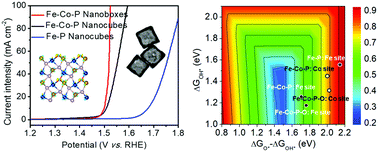
Efficient electrocatalysts are of great importance in improving the water splitting efficiency. Herein, we develop a self-templating strategy to construct porous iron cobalt (oxy)phosphide (Fe–Co–P) nanoboxes as promising pre-catalysts for the oxygen evolution reaction in alkaline solution. The constructed Fe–Co–P nanoboxes exhibit excellent electrocatalytic activity and afford a current density of 10 mA cm−2 at a small overpotential of 269 mV. Moreover, the structural evolution of the metal phosphides in the oxygen evolution process has been well monitored. X-ray absorption near-edge structure analyses and computational studies reveal that the structural merits and the effective intramolecular electronic coupling between the Fe and Co atoms via P/O bridges are responsible for the greatly improved electrocatalytic activity.


 Please wait while we load your content...
Please wait while we load your content...