Expanded hydrated vanadate for high-performance aqueous zinc-ion batteries†
Abstract
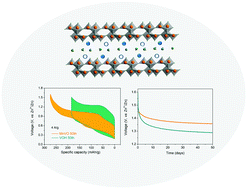
Hydrated vanadates are promising layered cathodes for aqueous zinc-ion batteries owing to their specific capacity as high as 400 mA h g−1; however, the structural instability causes serious cycling degradation through repeated intercalation/deintercalation reactions. This study reveals the chemically inserted Mn(II) cations act as structural pillars, expand the interplanar spacing, connect the adjacent layers and partially reduce pentavalent vanadium cations to tetravalent. The expanded interplanar spacing to 12.9 Å reduces electrostatic interactions, and transition metal cations collectively promote and catalyze fast and more zinc ion intercalation at higher discharge current densities with much enhanced reversibility and cycling stability. Manganese expanded hydrated vanadate (MnVO) delivers a specific capacity of 415 mA h g−1 at a current density of 50 mA g−1 and 260 mA h g−1 at 4 A g−1 with a capacity retention of 92% over 2000 cycles. The energy efficiency increases from 41% for hydrated vanadium pentoxide (VOH) to 70% for MnVO at 4 A g−1 and the open circuit voltage remains at 85% of the cutoff voltage in the MnVO battery on the shelf after 50 days. Expanded hydrated vanadate with other transition metal cations for high-performance aqueous zinc-ion batteries is also obtained, suggesting it is a general strategy for exploiting high-performance cathodes for multi-valent ion batteries.
- This article is part of the themed collection: 2019 Energy and Environmental Science HOT Articles


 Please wait while we load your content...
Please wait while we load your content...