Ethylene-glycol ligand environment facilitates highly efficient hydrogen evolution of Pt/CoP through proton concentration and hydrogen spillover†
Abstract
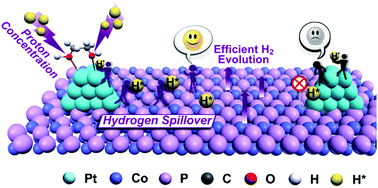
The low catalytic kinetics of many non-precious electrocatalysts for hydrogen evolution reaction is often associated with their adverse hydrogen adsorption/desorption kinetics. Thus, improving their kinetics as well as understanding the mechanism is critically important. Herein, the strategy of utilizing unique ethylene glycol ligand environments was employed to circumvent the aforesaid kinetic limitations in the composites of Pt-loaded CoP linked by ethylene-glycol through proton concentration and hydrogen spillover. At a low Pt loading of 1.5 wt%, the catalytic performance was significantly improved with dramatically decreased Tafel slopes from 104.6 mV dec−1 of CoP to 42.5 mV dec−1. Control experiments and theoretical calculations revealed that ethylene-glycol concentrated hydrogen intermediates at Pt (>7.3 times), facilitated the hydrogen spillover from hydrogen-enriched Pt to hydrogen-deficient CoP and modulated the local electronic structures to afford thermo-neutral Pt/CoP interfacial sites, improving the catalytic kinetics ultimately. The composites with 1.5 wt% Pt loading delivered a low overpotential of 21 mV at 10 mA cm−2 as well as record high noble-metal utilization activity, outperforming commercial Pt/C and other hydrogen spillover electrocatalysts for HER. This strategy may provide insights for the design of electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...